KF Spotlight – Global Market Reactions on COVID-19 Vaccine Development
Prepared by Aron Liew, CFA and Zainal Aiman
No one could have ever imagined that most of the year’s narrative will be about a microscopic parasite.
Story began when a strange new virus was found in Wuhan, China. The virus, named as COVID-19, which stands for Coronavirus Disease 2019, had spread swiftly across the globe including Malaysia, prompting the World Health Organization (WHO) to declare it as global pandemic. Given the rapid infection of the virus, many nations around the world decided to take unprecedented measures to contain the spread by imposing lockdown measures and border shutdown. As a result, most nations suffered steep economic slowdown.
Although countries around the world have resumed their economic activities since May 2020, work and interactions with other people have all been altered in the ‘new reality’. Face masks became a familiar sight, and social distancing became a new way of life.
Given that some section of economies cannot stay with this new normal forever, dozens of research teams around the world are working to develop a vaccine for this virus. Billion of dollars are invested in this development with countries like US, China and Europe investing the most. Recently, in November, positive news on the vaccine development by the manufacturers had sent global markets into a frenzy and providing a much-needed uplifting boost to a challenging year.
DO YOU KNOW?

The smallpox vaccine, introduced by Edward Jenner in 1796, was the first successful vaccine to be developed. He observed that milkmaids who previously had caught cowpox did not catch smallpox and showed that inoculated vaccinia protected against inoculated variola virus.
Source : WHO
What is vaccination?
According to WHO, vaccination is a simple, safe and effective way of protecting people against harmful diseases, before they come into contact with them. They train our immune system to create antibodies by killing or weakening viruses or bacteria, but they do not cause the diseases. Most vaccines are given by an injection, but some are given orally or sprayed into the nose.
How does a vaccine work?

What is in a vaccine?
Antigen
Kills or weakens the virus or bacteria and trains our bodies to recognize the disease in the future
Adjuvants
Help to boost our immune response which means to help vaccines to work better
Preservatives
Ensure a vaccine stays effective
Stabilisers
Protect the vaccine during storage and transportation.
Source : World Health Organisation
The Clinical Stage of Vaccine Production
Like all vaccines, COVID-19 vaccine will have to go through four (4) phases of clinical trials :
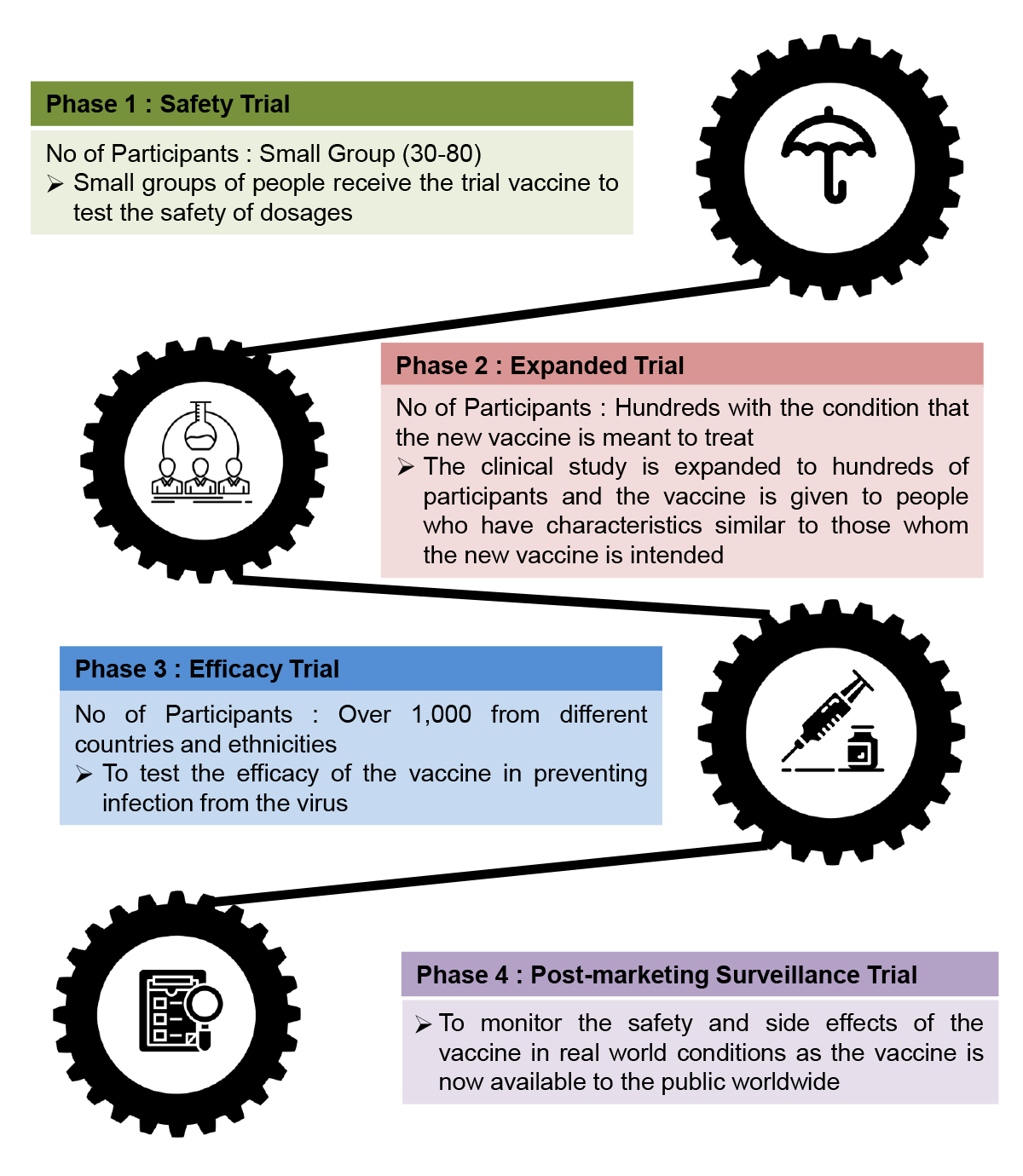
Source : Centers for Disease Control and Prevention
Update on COVID-19 Vaccine Development
According to WHO, as at 12 November 2020, there are 48 and 164 candidate vaccines in clinical and preclinical evaluation respectively. However, for the purpose of this report, we have shortlisted 4 candidate vaccines that are said to be near to completion.
| Name | Company | Progress | Latest Development |
| mRNA-1273 | Moderna Inc. | Phase 3 |
|
| BNT162b2 | Pfizer Inc, BioNTech SE and Fosun Pharma | Phase 3 |
|
| ChAdOx1nCov-19 | The University of Oxford and AstraZeneca PLC | Phase 3 |
|
| Sputnik V | Gamaleya Research Institute | Phase 3 |
|
Source : WHO, Bloomberg News and New York Times
Impact of Vaccine’s News on Major Stock Market
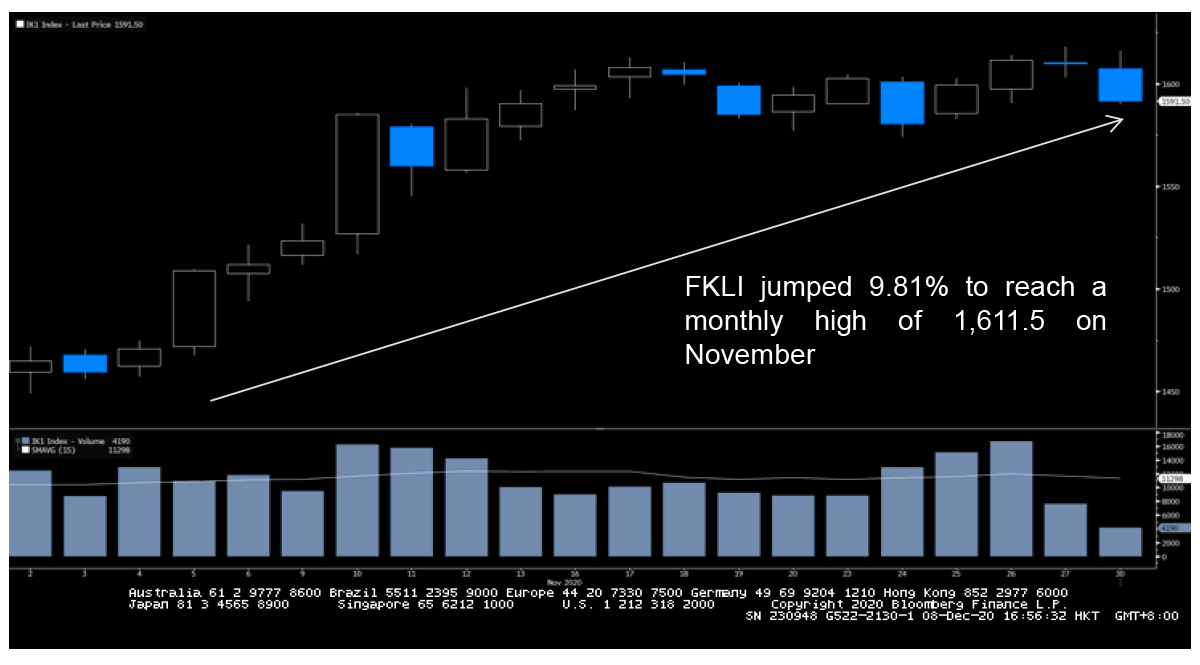
After all the relentless performance throughout the year, the month of November has at least provided some hopes to the stock market recovery. On top of the US Presidential announcement, the news on Vaccine Development has also boosted most of the major stock market’s performance in November, amid the hopes of potential global economic recovery. After the drug companies Pfizer and BioNTech announced the more than 90% effectiveness in their trials, most of the global stocks surged to record highs with the S&P 500 and FTSE 100 reaching 3,645.99 and 6,186 points, respectively. FBMKLCI also showed a positive reaction on November as the local bourse recorded its strongest performance in over two months to 1,575.07 on 10th November due to the news of COVID-19 vaccines
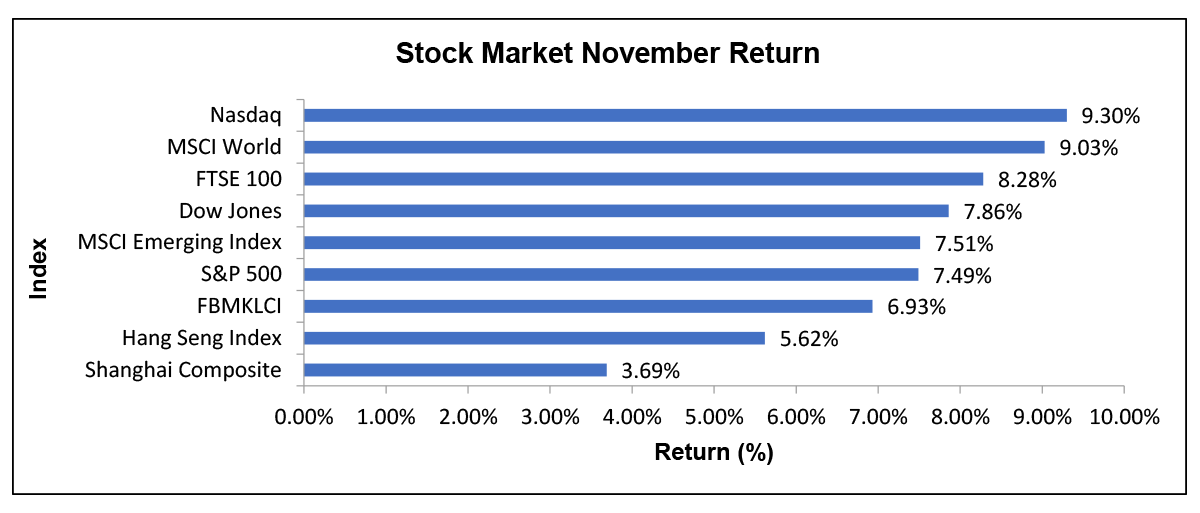
Impact of Vaccine’s News on Major Stock Market
Unlike the indices, commodity markets yielded mixed result amid the news of COVID-19 vaccines development. Gold and Silver showed negative readings on their November return. However, different performance were shown on other commodities with Brent and WTI crude oil showed impressive recoveries
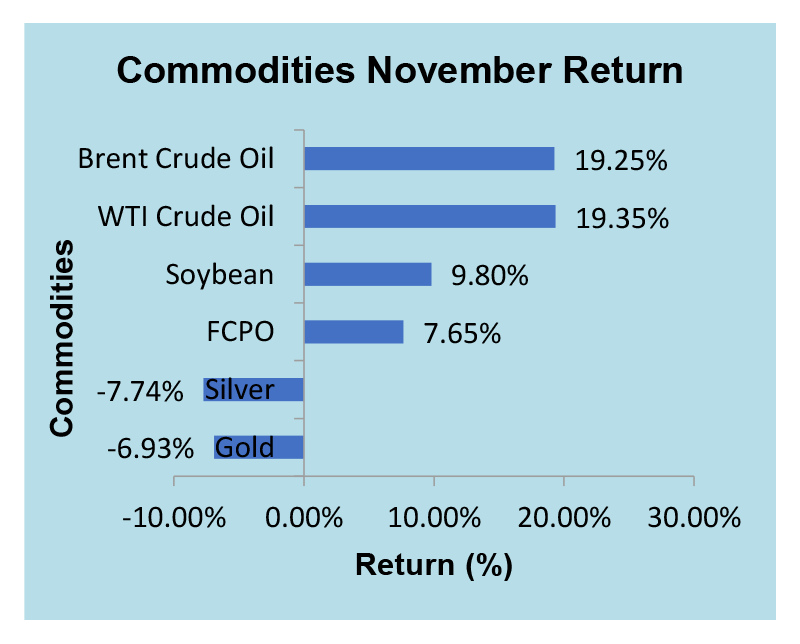
Source : Bloomberg
FKLI Movement on November
FCPO Movement on November
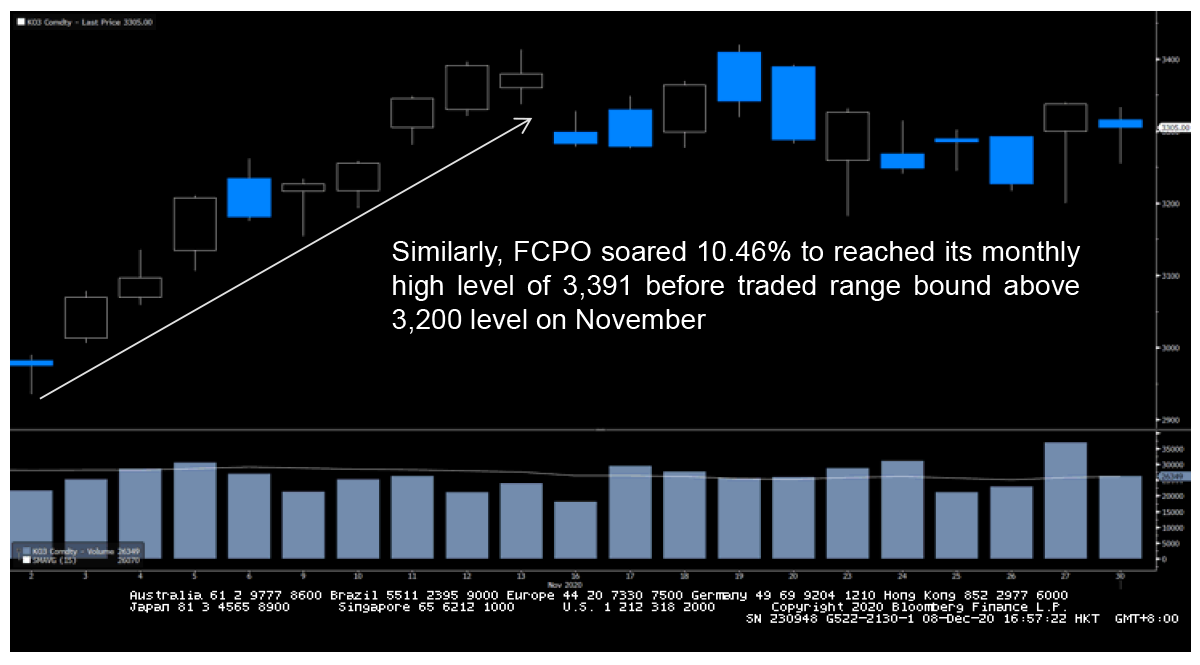
Source : Bloomberg
COVID-19 Vaccine Development in Malaysia
- Based on announcement made by Malaysian government on 27 November 2020, government is actively negotiating with 10 of the 12 companies whose COVID-19 vaccine is in the third phase of clinical study.
-
- So far, Malaysia government has inked two deals that would cover COVID-19 access to 30% of Malaysian population :
- A preliminary purchasing agreement has been made with Pfizer to obtain 12.8 million doses of COVID-19 vaccines to meet the immunization needs of 20% Malaysian population.
- A deal with the COVID-19 vaccine global access (COVAX) facility for 10% of Malaysians
- The vaccination process will be done in stages with priority will be given to high risk groups prone to COVID-19 infections including frontliners, senior citizens and patients with non-communicable diseases.
- On top of that, the government had also announced that the Ministry of Health would begin Phase 3 clinical research of the COVID-19 vaccine via a government-to-government initiative with China
Source : The Edge Market
Hedging Opportunities on Listed Derivatives
The news on COVID-19’s vaccine development has indeed spurred the recent rally on major stock markets and commodities around the world. Except for Gold and Silver, which currently experienced a downward movement in price, the rollout of positive vaccine development has instilled market optimism on potential global economic recovery next year. We expect this momentum to carry on until end of this year while safe haven commodities may not be as attractive.
However, the worst are not over yet. According to WHO, the widespread availability of any vaccines remain a long way off, while the cases and deaths keep increasing in many parts of the world. Some nations such as Hong Kong are also experiencing resurgence in cases. Global travel and trade will not see an immediate recovery while global demand and supply of commodities are still on the mend. After seeing a rebound from the news of the vaccine development, crude oil traders will focus their eyes on OPEC’s decision while equity index traders will continue to look at fiscal and monetary policies to support the economy.
Therefore, traders should still remain cautious amid the volatile market condition. Given this, listed derivatives offers an alternative opportunity to seek exposure or hedge one’s exposure in this market.
Download full article here
Disclaimer:
This document has been prepared solely for the use of the recipient. No part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means without the prior written permission from Kenanga Futures Sdn Bhd. Although care has been taken to ensure the accuracy of the information contained herein, Kenanga Futures Sdn Bhd does not warrant or represent expressly or impliedly as to the accuracy or completeness of the information. This information does not constitute financial or trading advice; neither does it make any recommendation regarding product(s) mentioned herein. Kenanga Futures Sdn Bhd does not accept any liability for any trading and financial decisions of the reader or third party on the basis of this information. All applicable laws, rules, and regulations, from local and foreign authorities, must be adhered to when accessing and trading on the respective markets.